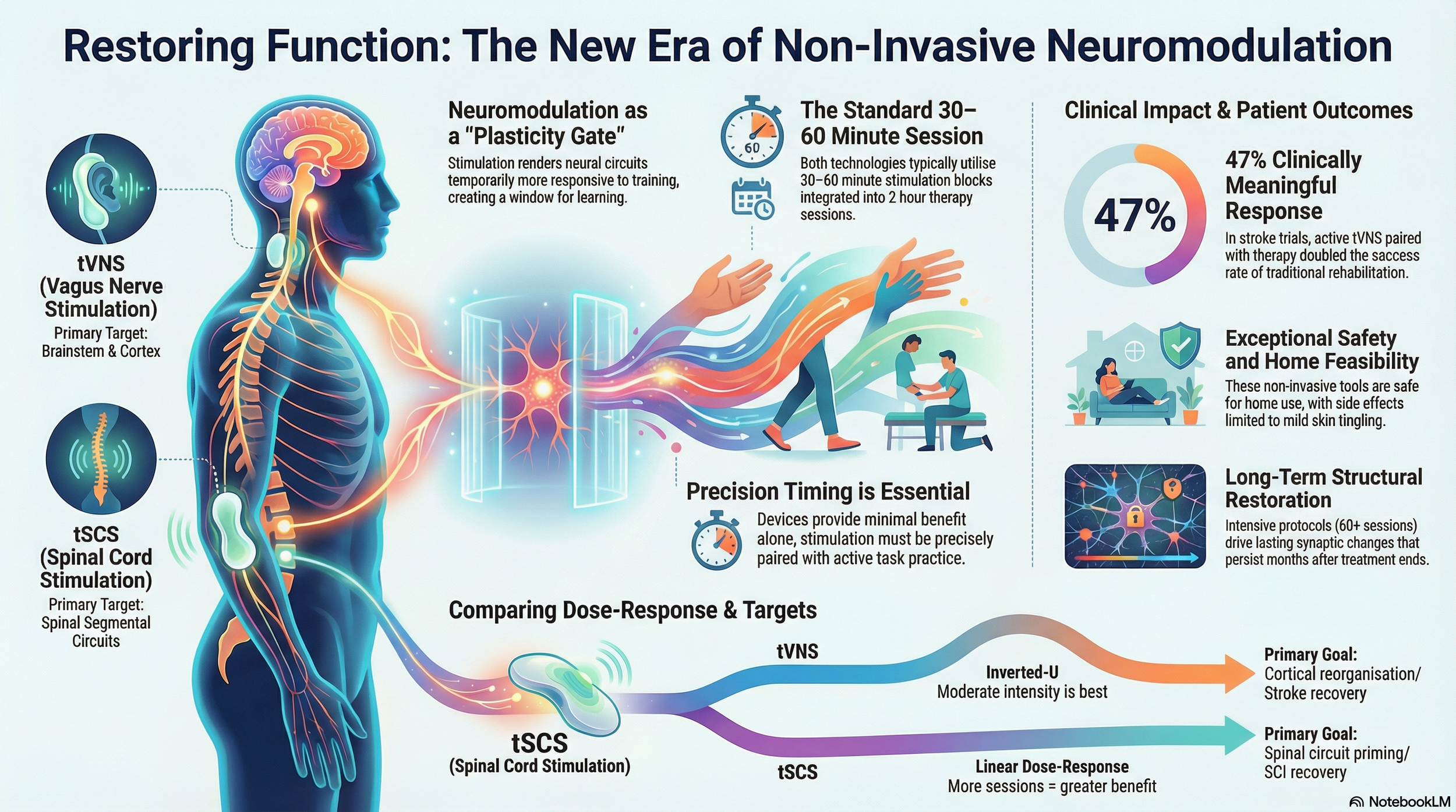
Transcutaneous Vagus Nerve Stimulation and Transcutaneous Spinal Cord Stimulation - Parallels for Rehabilitation Practice
Two non-invasive neuromodulation technologies — transcutaneous vagus nerve stimulation (tVNS) and transcutaneous spinal cord stimulation (tSCS) — are reshaping how we think about neurological rehabilitation. Although they target different levels of the nervous system, these modalities share a surprising number of fundamental principles. Understanding these parallels gives clinicians, patients, and carers a clearer picture of how modern neuromodulation works, what to expect from treatment, and why these technologies represent a genuine shift from compensatory to restorative rehabilitation.
This article identifies ten fundamental parallels between tVNS and tSCS, drawing on the published scientific evidence to explore what they have in common, where they differ, and what this means for clinical practice.
1. Timing-Dependent Paired Rehabilitation: The Critical Synchronisation Principle
The most profound parallel between tVNS and tSCS lies in their shared reliance on precision timing to drive neuroplastic reorganisation. Neither modality functions effectively as a standalone stimulation; both require temporally synchronised pairing with active task practice to produce functional gains.
Mechanistic Foundation
For tVNS, the critical timing window was elegantly demonstrated in rat stroke models: vagus nerve stimulation paired during forelimb movement produces significant functional recovery, whereas stimulation delivered after movement completion fails to enhance training (Hays, 2016).
The mechanism involves tVNS-triggered release of neuromodulators — norepinephrine from the locus coeruleus and acetylcholine from the nucleus basalis — that must coincide temporally with task-relevant neural activity to strengthen functionally useful connections. This timing-dependent mechanism mirrors the principles of spike-timing-dependent plasticity (STDP): when neuromodulator release overlaps with movement-related neural firing, the salience of that movement increases, promoting clinically useful neural reorganisation (Brzosko et al., 2019; Feldman, 2012).
Similarly, tSCS creates a neuroplastic window by transiently enhancing spinal inhibitory circuits and modulating afferent input to the spinal cord. The stimulation must be delivered concurrently with voluntary motor attempts or assisted movements to capitalise on the enhanced circuit excitability. When tSCS-evoked afferent volleys arrive at spinal circuits simultaneously with descending motor commands, activity-dependent strengthening of relevant synaptic connections occurs (Evancho et al., 2023; Hofstoetter et al., 2024; Iddings et al., 2021).
Clinical Translation
The VNS-REHAB pivotal trial for stroke upper limb recovery exemplifies optimal implementation: participants received 0.5-second bursts of VNS precisely synchronised with each successful movement during task-specific training — reaching for objects, grasping, manipulating — totalling 400–600 movement-VNS pairs per 2-hour therapy session. The control group received identical therapy with subthreshold VNS, isolating the plasticity-enhancing effect of properly timed stimulation. Results demonstrated a mean 5.0-point improvement in the Fugl-Meyer Upper Extremity (FMA-UE) assessment for the active VNS group compared with 2.4 points for control therapy (p<0.001), with a 47% clinically meaningful response rate (≥6-point improvement) versus 24% in controls (Dawson et al., 2021).
For tSCS, the Up-LIFT trial employed 30 minutes of cervical stimulation concurrent with upper extremity task practice, followed by 60–90 minutes of continued training without stimulation. This protocol leverages the 2-hour carryover window during which spinal circuits remain modulated, allowing extended practice under favourable plasticity conditions without continuous stimulation (Moritz et al., 2024; Hofstoetter et al., 2020).
What This Means in Practice
Both modalities require structured therapy sessions where stimulation timing is coordinated with specific movements or tasks. Using a standalone device without paired training yields minimal functional benefit. Training protocols should emphasise repetitive-task practice (400–600 repetitions per session for upper limb; 1,000+ steps for gait training), with stimulation delivered either concurrently (tSCS) or at movement initiation (tVNS). As Hays et al. (2023) put it in their appropriately titled paper, "How to fail with paired VNS therapy," insufficient training intensity or poor timing are the most reliable ways to fail to see benefit from neuromodulation.
2. Session Duration and Frequency: Converging on 30–60 Minute Protocols
Despite targeting different neural levels — tVNS acting supraspinally via brainstem nuclei, tSCS acting spinally via afferent pathways — both modalities have converged on remarkably similar session parameters through independent clinical optimisation.
Standard Session Architecture
For tVNS, comprehensive safety analyses across 60+ studies involving over 6,000 participants establish that 30, 60minute sessions at 25–50% duty cycle (stimulation on:off ratio) are safe and free of serious adverse events when repeated with ≥12–24-hour intervals. The VNS-REHAB protocol employs 2-hour sessions, with tVNS delivered as 0.5-second bursts at each movement, resulting in a cumulative stimulation time of approximately 3–5 minutes distributed across the session. Alternative protocols for autonomic modulation or migraine use continuous 15–60 minute sessions (Kampusch et al., 2022; Redgrave et al., 2018; Dawson et al., 2021).
For tSCS, the field has standardised on 30–60 minute continuous stimulation sessions, typically integrated with 60–90 minutes of rehabilitation. Session frequency ranges from 2 to 5 times per week, with 3 times weekly emerging as the evidence-based standard for long-duration protocols (Moritz et al., 2024; Hofstoetter et al., 2020; Hofstoetter et al., 2021).
Dose-Time Relationships
The temporal patterning differs meaningfully: tVNS often uses intermittent bursts (30 seconds on, 30 seconds off for auricular; 0.5-second pulses synchronised with movement for paired rehabilitation), whereas tSCS typically delivers continuous stimulation throughout the 30-minute window. This difference likely reflects the distinct mechanisms — tVNS activates central neuromodulatory nuclei that remain active for minutes after stimulation ceases, whereas tSCS directly modulates spinal circuit excitability, requiring sustained input (Butt et al., 2020; Yaghouby et al., 2024; Dawson et al., 2021).
However, both show evidence of "minimum effective dose" thresholds below which benefits diminish. For tVNS in perceptual decision-making, 4-second stimulation trains reliably increased locus coeruleus activity (indexed by pupil dilation) lasting 5.8 seconds post-stimulation, but shorter trains produced inconsistent effects (D'Agostini et al., 2024). For tSCS, pilot data suggest that 15-minute sessions may be insufficient to produce the 2-hour carryover effects observed with 30-minute protocols (Hofstoetter et al., 2020).
What This Means in Practice
Both modalities fit within standard 1–2 hour therapy blocks common in NHS and private rehabilitation settings. The 30-minute stimulation plus 60 to 90-minute continued practice model translates readily to existing clinical workflows without requiring restructuring of therapy schedules.
3. Dose-Response Characteristics: Linear vs. Non-Monotonic Curves
A critical distinction with important clinical implications emerges in dose-response relationships. While both modalities show parameter-specific optimisation, tSCS demonstrates generally linear dose-response (more sessions = greater benefit, up to limits), whereas tVNS exhibits an inverted-U, non-monotonic dose-response (moderate intensity is optimal).
tVNS: The Inverted-U Phenomenon
Elegant preclinical work by Pruitt et al. (2020) demonstrated that moderate-intensity VNS (0.8 mA) paired with rehabilitative training significantly enhanced forelimb recovery after ischaemic stroke in rats, whereas both lower (0.4 mA) and higher (1.6 mA) intensities failed to improve recovery compared with rehabilitation without VNS. The inverted-U occurs because VNS intensity determines locus coeruleus spike activity in a non-linear fashion: moderate intensities produce optimal norepinephrine release patterns, but excessive intensities may trigger competing inhibitory mechanisms or overwhelm the system.
Human optimisation studies corroborate this finding. In veterans with PTSD, the parameter combination of 20 Hz frequency, 100 µs pulse width, and 80% discomfort threshold produced superior outcomes (improved slow-wave sleep, reduced startle response) compared to 100% sensory threshold or 200% sensory threshold intensities. Critically, the 80% discomfort threshold setting allowed higher absolute current delivery while maintaining tolerability, suggesting the sweet spot balances physiological activation with comfort (Strawn et al., 2025).
The non-monotonic dose-response has profound clinical implications: "more stimulation does not necessarily produce greater benefits," requiring clinicians to consider both increasing *and* decreasing parameters for non-responders. This contrasts sharply with pharmacological paradigms, where dose escalation is standard for insufficient response (Pruitt et al., 2020).
tSCS: Cumulative Session Benefits
tSCS dose-response appears fundamentally different: there is a clear threshold of 60 sessions minimum for sustained functional improvements in chronic spinal cord injury, with continued gains beyond 60 sessions and no apparent plateau through 120 sessions. A year-long pilot study documented that "slow and gradual improvements in outcome measures continued to be noted over the 120 sessions, which did not seem to plateau at the conclusion of the study" (Phillips et al., 2025). Post-hoc analyses from a multicentre trial confirmed that improvements required ≥60 tSCS-activity-based therapy sessions, with larger effect sizes as session counts increased (Moritz et al., 2024).
For tSCS intensity parameters (stimulation amplitude), the relationship appears more linear: higher amplitudes within tolerance generally produce stronger effects, with individuals having ASIA Impairment Scale B (motor-complete) injuries requiring significantly higher amplitudes than AIS C-D (incomplete) to achieve equivalent motor responses (Tefertiller et al., 2024).
Mechanistic Interpretation
These contrasting dose-response patterns likely reflect the different neuroplastic mechanisms:
tVNS modulates central neuromodulatory tone (LC-NE system), which follows homeostatic regulatory principles. Excessive NE release may activate compensatory downregulation or engage inhibitory β-adrenergic mechanisms (Pruitt et al., 2020).
tSCS drives structural synaptic changes (KCC2 membrane expression restoration, altered chloride homeostasis, modified H-reflex pathways) that accumulate incrementally with repeated sessions, similar to motor learning requiring distributed practice over weeks to months (Malloy & Cote, 2024).
What This Means in Practice
For tVNS, clinicians should begin at moderate intensities (80% discomfort threshold or maximum comfortable), carefully titrate, and recognise that non-responders may require parameter reduction rather than escalation. The assumption that "more is better" does not apply.
For tSCS, patients should understand that a minimum of 20 sessions (for modest gains in subacute populations) and ideally 60+ sessions are needed for robust functional restoration in chronic SCI. The good news is that continued improvements beyond initial treatment blocks justify ongoing rehabilitation programmes.
4. Multi-Tiered Carryover Effects: From Hours to Months
Both modalities produce temporally layered carryover effects — the persistence of therapeutic benefits after stimulation ceases — which is critical for determining optimal treatment schedules and setting realistic outcome expectations.
Immediate Carryover (Minutes to Hours)
For tVNS, 4-second stimulation trains reliably increase pupil dilation (indexing LC-NE activity) that persists for approximately 5–6 seconds post-stimulation, extending through most of the decision-making period in perceptual tasks, even though stimulation ended earlier. In cognitive performance studies, cervical tVNS improved objective arousal and multitasking for up to 24 hours post-stimulation in sleep-deprived individuals (D'Agostini et al., 2024; Svensson et al., 2021).
For tSCS, the 2-hour carryover window is well characterised mechanistically: a single 30-minute session produces measurable improvements in walking performance, postural control, and spasticity measures that persist for 2 hours but return to baseline by 24 hours (except for Modified Ashworth Scale muscle tone, which remains improved at 24 hours). Neurophysiological investigations reveal that postsynaptic reciprocal Ia inhibition and presynaptic inhibition remain enhanced during the first 3–75 minutes post-tSCS, returning to baseline by 120–190 minutes — directly explaining the 2-hour functional window (Hofstoetter et al., 2020; Hofstoetter et al., 2024).
Extended Carryover (Days to Weeks)
Published data on week-scale carryover remain limited for both modalities but suggest meaningful persistence after multi-session protocols. For tVNS in progressive MS, effects on spasticity persisted at 1-week follow-up after 4 weeks of treatment (8 sessions). For tSCS, cardiovascular improvements in chronic cervical SCI persisted "for several weeks" after six 30-minute sessions over 2 weeks, though effects had diminished by the 6-week assessment. A targeted cervical tSCS protocol included a 3-week "No Stim" washout period after 16 weeks of continuous weekly stimulation; notably, participant gains persisted during this month-long period without stimulation (Hofstoetter et al., 2021; Inanici et al., 2021).
Long-Term Neuroplastic Persistence (Months)
The most compelling evidence for sustained neuroplastic reorganisation comes from case reports and pilot studies. A participant with chronic cervical SCI (C3, 8 months post-injury) underwent approximately 4–5 weeks of tSCS combined with physical therapy. Following this intervention, the neurological level of injury improved from C3 to C4 and was sustained for 3 months of follow-up with no additional stimulation or therapy. The participant resumed self-feeding for the first time since the injury, with functional improvements persisting throughout the follow-up period. This duration exceeds natural recovery expectations in chronic SCI, suggesting activity-dependent structural reorganisation (Inanici et al., 2020).
Animal models provide mechanistic insight: rats receiving 18 sessions of tSCS over 6 weeks (3x/week) demonstrated sustained restoration of KCC2 membrane expression on lumbar motoneurons — a structural change underlying normalised chloride homeostasis and reduced hyperreflexia. These molecular-level changes persisted at the 6-week endpoint, supporting the biological plausibility of months-long carryover (Malloy & Cote, 2024).
What This Means in Practice
The tiered carryover architecture informs treatment frequency decisions. Acute symptom management (e.g., reducing spasticity before a therapy session) can use single applications producing 2–24 hour benefits. Subacute functional restoration typically requires 2–5 sessions per week over 4–8 week blocks. Long-term maintenance schedules, after achieving gains through intensive protocols, may be sustainable at reduced frequencies, though systematic data remain limited.
5. Neuromodulation as Plasticity Primer: Shared "Gating" Mechanisms
Perhaps the most conceptually important parallel lies in recognising both tVNS and tSCS as plasticity primers or gates rather than direct functional effectors. Neither produces sustained functional improvement when applied without training; instead, they transiently create neurobiological conditions favourable for activity-dependent learning.
tVNS: Gating Cortical Plasticity via Neuromodulatory Nuclei
tVNS activates the vagus nerve's auricular or cervical branch, triggering ascending projections to the nucleus tractus solitarius in the brainstem, which then activates the locus coeruleus-norepinephrine (LC-NE) system and nucleus basalis cholinergic system. These neuromodulatory nuclei innervate the entire cortex, releasing norepinephrine and acetylcholine that modulate cortical excitability and synaptic plasticity (Butt et al., 2020; Baig et al., 2022).
Critically, this neuromodulation is state-dependent:
Norepinephrine and acetylcholine release timed with task-relevant neural activity strengthen active synapses according to Hebbian principles ("neurons that fire together, wire together").
Recent evidence demonstrates that tVNS directly increases pupil-indexed LC-NE activity and enhances accuracy in perceptual decision-making without affecting reaction time — indicating improved evidence accumulation rather than urgency or speed-accuracy tradeoffs. The effect was selective: tVNS prevented post-error accuracy declines in less proficient participants but had no effect in high-performers or after correct trials, demonstrating context-sensitivity (D'Agostini et al., 2024).
Furthermore, tVNS increases brain-derived neurotrophic factor (BDNF) levels — a neurotrophin essential for long-term potentiation, synaptic remodelling, and motor learning consolidation. By elevating tonic BDNF availability while task-specific training recruits particular motor networks, tVNS may preferentially strengthen those task-relevant connections (Baig et al., 2022; Dawson et al., 2021).
tSCS: Gating Spinal Plasticity via Afferent Modulation
tSCS achieves analogous gating at the spinal level by repetitively activating large-diameter proprioceptive and cutaneous afferents, transiently enhancing spinal inhibitory circuits and modulating motoneuron pool excitability. This creates a window during which voluntary motor commands engage spinal circuitry closer to normative function — reduced hyperreflexia, enhanced reciprocal inhibition, and improved frequency-dependent modulation (Hofstoetter et al., 2024; Malloy & Cote, 2024).
The critical insight from animal models is that multi-session tSCS mimics aspects of motor training by engaging activity-dependent plasticity mechanisms. Repeated afferent activation over weeks prevents the SCI-induced downregulation of KCC2 membrane expression, maintaining the chloride gradient necessary for effective GABAergic inhibition. This represents a structural synaptic modification that requires accumulated exposure, explaining why single sessions produce transient effects while 60+ sessions yield persistent reorganisation (Malloy & Cote, 2024; Malloy & Cote, 2025).
The "Priming" Paradigm
Both modalities exemplify priming-based rehabilitation: brief neuromodulation renders neural circuits temporarily more responsive to training, allowing clinicians to "open the plasticity gate" and maximise training efficacy during that window. This contrasts with substitutive approaches (orthotics, functional electrical stimulation producing movement directly) or compensatory approaches (training alternative movement strategies) (Evancho et al., 2023; Iddings et al., 2021).
A 2023 review of combined neuromodulation and physical therapy concluded that "neuromodulatory stimulation approaches activate many of the same circuits that are activated by training at all levels of the neuraxis, and therefore have the potential to reverse maladaptive reorganisation" (Evancho et al., 2023). The review emphasises that combinatorial approaches — neuromodulation plus intensive task-specific training — are most likely to produce meaningful functional restoration.
What This Means in Practice
Both tVNS and tSCS are training enhancers, not passive treatments. Optimal outcomes require commitment to intensive concurrent rehabilitation. Stimulation without training provides minimal benefit — the neuromodulation opens a window, but only purposeful practice drives functional reorganisation through that window. This is an important message for anyone considering these therapies: the device alone is not the treatment. The device paired with hard work is.
6. Parameter Complexity and Individualisation Requirements
Both modalities involve multidimensional parameter spaces that require individualised optimisation, presenting implementation challenges that demand clinical expertise.
tVNS Parameter Landscape
Key adjustable parameters include (Butt et al., 2020; Farmer et al., 2021):
- Stimulation site: Auricular (tragus, cymba conchae) vs. cervical (carotid region)
- Frequency: 10–50 Hz (20–30 Hz most common; higher frequencies for antinociceptive effects)
- Pulse width: 100–500 µs (shorter durations tolerate higher intensities)
- Intensity: Individual sensory/discomfort threshold (typically 2–6 mA for auricular, 12–14 V for cervical devices)
- Duty cycle: Continuous vs. intermittent (e.g., 30s on/30s off)
- Session duration: 15–60 minutes
- Laterality: Left-sided most common (clinical convention based on cardiac safety considerations)
Optimisation studies demonstrate non-obvious interactions: the 20 Hz, 100 µs, 80% discomfort threshold combination outperformed other frequency-pulse width-intensity combinations in PTSD, with the shorter pulse width allowing higher intensity delivery while maintaining tolerability (Strawn et al., 2025).
tSCS Parameter Landscape
Adjustable parameters include (Tefertiller et al., 2024):
- Electrode positioning: Cathodal electrode at target spinal level (C3-C7 for upper extremity, T11-L2 for lower extremity); anodal electrodes bilaterally at collar bones, abdomen or pelvis
- Stimulation amplitude: Individual motor threshold (50–150 mA typical range; AIS B injuries require higher amplitudes than AIS C-D)
- Frequency: 30–50 Hz (30 Hz standard, 50 Hz for anti-spasticity applications)
- Pulse width: 1 ms per phase
- Carrier frequency: 5–10 kHz (enhances comfort, reduces skin sensation — not essential and not deployed in most systems)
- Waveform: Monophasic vs. biphasic (83% of sessions use biphasic)
A framework developed from 77 participants across two large SCI trials proposes a hierarchical adjustment sequence: (1) gradually increase current amplitude to achieve motor effects or maximum tolerance, (2) select waveform type based on tolerability, (3) optimise electrode positioning for the target spinal level, (4) adjust the burst frequency based on the application (30 Hz for motor, 50 Hz for spasticity). Device-related adverse events were infrequent and not correlated with specific waveforms or amplitudes, supporting the safety of this optimisation process (Tefertiller et al., 2024).
Shared Challenge: Responder Heterogeneity
A fundamental challenge in transcutaneous neuromodulation is substantial interindividual variability in response, even within well-defined patient populations. The EChO study of implanted spinal cord stimulation documented "remarkably large interindividual variation" in carryover duration (median 5 hours, interquartile range 2.5–21 hours) — likely applicable to tSCS given similar mechanisms (Meier et al., 2024). For peripheral nerve stimulation, baseline psychological resilience (pain self-efficacy, pain catastrophising) and physical activity levels emerged as the strongest predictors of treatment response (Gilligan et al., 2023).
Currently, neither tVNS nor tSCS has validated biomarkers for prospectively identifying responders before initiating treatment, though some trends exist:
tSCS responders: Incomplete injuries (AIS B-D) respond better than complete injuries; moderate disability levels benefit most (MS patients requiring walker/crutches improved more than those with very low or very high function) (Phillips et al., 2025; Hofstoetter et al., 2020).
tVNS responders: Limited data; one peripheral nerve stimulation study found that TENS-positive patients had 94% positive response to permanent nerve stimulation, suggesting trial periods with simpler devices may predict advanced neuromodulation success (Deer et al., 2020).
What This Means in Practice
Both modalities require clinicians capable of systematic parameter optimisation based on individual response. Protocols should include initial 2–3 session titration periods with documented parameter adjustments and functional outcome tracking (e.g., 10-metre walk test, Box and Blocks, spasticity scales). Structured trial protocols — single-session assessments with pre- and post-outcome measures — can help identify non-responders early and avoid extended, ineffective treatment.
7. Home-Based Feasibility and Telehealth Compatibility
A compelling practical parallel lies in both modalities' successful translation to remotely supervised, self-administered home therapy — critical for scaling access beyond specialist clinics and supporting long-duration treatment protocols.
tVNS Home Implementation Evidence
Multiple rigorous studies demonstrate the safety and feasibility of home-based tVNS. A 2022 pilot randomised controlled trial examined fully contactless, remotely supervised self-administered auricular tVNS in 12 participants with Long COVID. Participants self-administered 1-hour sessions twice daily, six days per week, for 4 weeks (total 56 sessions) (Badran et al., 2022).
Key findings:
- Rapid competence acquisition: At initial pre-stimulation orientation, 91% required assistance with heart rate monitoring and 100% required assistance with tVNS setup. By the 5th session (day 3), 100% of users self-administered all procedures without assistance.
- Exceptional compliance: All 12 participants completed all required visits without dropout.
- Safety: No bradycardia or serious adverse events occurred during any of the 672 home sessions.
- Remote supervision effectiveness: Clinical staff supervised via videoconference, providing real-time guidance initially but minimal intervention by week 2.
Similar success was demonstrated in paediatric autism spectrum disorder: caregivers administered nightly 60-minute tVNS sessions at home under remote supervision, achieving 88.5% compliance. Even in this population with sensory hypersensitivities, tolerability was high and side effects minimal across 132 sessions (Badran et al., 2023).
tSCS Home Implementation Evidence
The Up-LIFT trial's follow-on "LIFT Home" study enrolled 17 participants with chronic cervical SCI who successfully transitioned to home-based self-administration after initial in-clinic training. Critical to home feasibility, participants maintained stimulation amplitudes within 1% of preset values, demonstrating reliable device operation without real-time clinical supervision. The study documented that, with thorough in-clinic programming and training, individuals with SCI (including those with upper-extremity impairments) could independently set up electrodes, activate devices, and execute paired training protocols at home (Tefertiller et al., 2025).
Enabling Factors for Home Translation
Several factors enable home-based neuromodulation across both modalities:
1. Non-invasive surface application: Unlike implanted systems requiring surgical expertise, transcutaneous approaches use simple electrode placement techniques learnable within 1–5 sessions (Badran et al., 2022; Badran et al., 2023).
2. Portable, user-friendly devices: Contemporary tVNS and tSCS devices are battery-powered, lightweight, and feature simplified interfaces suitable for patient/caregiver operation.
3. Telehealth supervision infrastructure: Videoconference platforms enable real-time observation during initial sessions, parameter adjustment, troubleshooting, and compliance monitoring without in-person visits.
4. Established safety profiles: Both modalities' excellent safety records and absence of serious device-related adverse events justify unsupervised home use after appropriate training (Kampusch et al., 2022; Redgrave et al., 2018; Tefertiller et al., 2024).
A Practical Clinical Workflow
A hybrid clinic-to-home model emerges as both practical and optimal:
- Weeks 1–2 (in-clinic): Initial evaluation, parameter optimisation, supervised pairing with task-specific training, caregiver training if applicable, demonstration of competence
- Weeks 3–8 (home-based): Self-administered sessions 2–5 times weekly with weekly telehealth check-ins for compliance monitoring, parameter adjustments, outcome tracking
- Month 3+ (maintenance): Reduced frequency home-based sessions (1–3 times weekly) with monthly telehealth follow-up
What This Means in Practice
Home-based delivery extends access beyond clinic capacity while enabling the extended treatment protocols (60+ sessions) that the evidence says are needed for robust functional restoration. For patients who live far from specialist centres, or who need long-duration protocols that would be impractical to deliver entirely in-clinic, the ability to transition to supervised home use is a significant advantage.
8. Exceptional Safety Profiles with Minimal Side Effects
A reassuring parallel for clinical implementation and patient counselling is that both modalities demonstrate excellent safety profiles across thousands of participants and hundreds of thousands of stimulation sessions, with adverse events predominantly mild, transient, and manageable through parameter adjustment.
tVNS Safety Evidence
The most comprehensive safety analysis to date pooled data from 177 studies comprising 6,322 participants receiving tVNS. Key findings (Kampusch et al., 2022):
- Overall adverse event rate: 12.84 events per 100,000 person-minutes-days of stimulation (95% CI 6.65–24.80)
- Most common side effects: Local skin irritation from electrode placement (18.2%), headache (3.6%), nasopharyngitis (1.7%)
- Dropout rate: Only 2.6% discontinued participation due to side effects
- Serious adverse events: 30 SAEs occurred across all studies, but only 3 were assessed by researchers as possibly caused by tVNS
A separate systematic review concluded that tVNS is "safe and well tolerated, with only mild side effects such as local skin irritation due to electrode placement, headache, and nasopharyngitis." Stimulation-related parameters (frequency, pulse width, intensity, duration, number of sessions) showed no correlation with adverse event rates, suggesting a wide therapeutic window (Redgrave et al., 2018).
Notably, transcutaneous approaches avoid the surgery-related adverse events associated with implanted VNS systems (infection rates 3–6%, cardiac events, hoarseness in 53.8% of implanted patients), representing a substantial safety advantage for non-invasive variants (Englot et al., 2016).
tSCS Safety Evidence
Safety data from large-scale tSCS trials corroborate the favourable profile observed with tVNS. The Up-LIFT framework study analysed 77 participants across two trials receiving cervical tSCS for upper extremity rehabilitation after chronic SCI. Device-related adverse events were infrequent and not correlated with specific waveforms or stimulation amplitudes, supporting the safety of the hierarchical parameter optimisation approach (Tefertiller et al., 2024).
Common tSCS side effects include:
- Local skin sensation: Tingling, prickling at electrode sites (typically adapts within first few sessions)
- Muscle contractions: Rhythmic paraspinal muscle contractions at stimulation frequency (can be an expected physiological response, not adverse)
- Transient discomfort: Occasionally reported during initial amplitude escalation (managed by reducing intensity or adjusting electrode position)
Importantly, no serious adverse events attributable to tSCS stimulation have been reported in major clinical trials involving hundreds of participants across thousands of sessions. The modality's safety profile enabled paediatric application and home-based use in individuals with substantial neurological impairments (Phillips et al., 2025; Moritz et al., 2024; Tefertiller et al., 2024).
Comparative Context
The safety profiles of tVNS and tSCS compare favourably to alternative rehabilitation interventions:
- Pharmacological spasticity management (baclofen, tizanidine): Sedation, weakness, hepatotoxicity risks requiring monitoring
- Botulinum toxin injections: Systemic spread, dysphagia, respiratory complications (rare but serious)
- Implanted spinal cord stimulation: Surgical risks, infection (3–8%), lead migration, hardware failure
- Standard intensive rehabilitation: Musculoskeletal overuse injuries, fatigue, cardiovascular stress in deconditioned populations
By contrast, tVNS and tSCS offer neuromodulation benefits with minimal systemic effects and reversible, user-controlled application. For patients and clinicians weighing the risk-benefit ratio, this is an important consideration.
9. Clinical Application Convergence: Upper Limb Rehabilitation
Beyond mechanistic parallels, both modalities have demonstrated clinical efficacy in overlapping functional domains, particularly in upper extremity rehabilitation — a high-priority goal with substantial impact on independence and quality of life.
tVNS for Stroke Upper Limb Recovery
The VNS-REHAB pivotal trial provides the most rigorous evidence for tVNS-enhanced upper-limb rehabilitation. This multi-site, double-blind, randomised trial enrolled 120 participants with upper limb weakness 9 months to 10 years after ischaemic stroke (mean time since stroke ~2 years). Participants received implanted VNS devices and were randomised to active VNS or sub-threshold (control) VNS, paired with identical task-specific upper limb rehabilitation (Dawson et al., 2021).
Treatment protocol: 18 outpatient therapy sessions over 6 weeks (3 sessions/week), each consisting of 90 minutes of intense task-specific training with 300–600 movement-VNS pairs per session, followed by a home exercise programme with VNS for 90 days.
Results at Day 1 after completion of 6-week in-clinic therapy:
- Primary outcome (FMA-UE change): 5.0 points (SD 4.4) in VNS group vs. 2.4 points (SD 4.4) in control group (p<0.001)
- Response rate (≥6-point improvement): 47% VNS vs. 24% control (odds ratio 2.8, p=0.001)
- Clinically meaningful improvement (≥6 points): Achieved by nearly twice as many VNS participants
These results are particularly striking because participants averaged 2 years post-stroke — well beyond the traditional window for spontaneous motor recovery.
tSCS for SCI Upper Limb Recovery
Cervical tSCS for upper extremity function after SCI demonstrated comparable efficacy. The Up-LIFT trial enrolled 60 participants with chronic cervical SCI (C1-C7 injuries, >6 months post-injury) who received 2 months of cervical tSCS paired with task-specific upper extremity training. Treatment occurred 3–5 times per week for 30 minutes, concurrent with reaching, grasping, and manipulation tasks, followed by continued training (Moritz et al., 2024).
Results showed that 72% of participants experienced meaningful improvements in arm and hand strength and function, with investigators noting that "many people were still improving at the end" of the 2-month period. Specific functional gains included increased grip strength, improved ability to manipulate objects, and enhanced independence in activities of daily living.
A case series of targeted cervical tSCS (C3-C7) in two individuals with motor-complete SCI (AIS A) employed 35 weeks of weekly sessions with task-specific training. Despite motor-complete classifications (no volitional movement below injury level), participants demonstrated improved sensation and proprioception in previously insensate dermatomes, enhanced voluntary muscle activation capacity, and functional gains that persisted after a 3-week "No Stim" washout period, suggesting lasting neuroplastic reorganisation (Inanici et al., 2021).
Mechanistic Convergence Despite Different Neural Targets
The parallel efficacy in upper limb rehabilitation despite different neuromodulatory targets — tVNS acting cortically via brainstem nuclei, tSCS acting spinally via segmental circuits — highlights that upper extremity function requires coordinated activity across multiple neural levels. Both interventions enhance activity-dependent plasticity within their respective target circuits:
- tVNS: Strengthens cortical motor representations, enhances corticospinal connectivity, promotes cortical map reorganisation (Dawson et al., 2021; Baig et al., 2022)
- tSCS: Normalises spinal reflex circuits, enhances motoneuron pool recruitment, improves sensorimotor integration (Moritz et al., 2024; Inanici et al., 2021)
The shared clinical outcome — improved upper extremity function — emerges from priming different components of the distributed sensorimotor network responsible for skilled arm and hand control.
What This Means in Practice
For upper extremity rehabilitation, patient selection may be guided by the neural level of impairment: predominantly cortical/subcortical lesions (stroke, traumatic brain injury) may favour tVNS, whereas spinal lesions (SCI, myelopathy) may favour tSCS. However, overlap exists, and some patients may benefit from both modalities targeting different levels of the motor system.
10. Integration with Rehabilitation Philosophy: From Compensation to Restoration
The final and perhaps most transformative parallel is that both tVNS and tSCS represent a fundamental shift in rehabilitation philosophy — from teaching compensatory strategies to promoting genuine functional restoration through neuroplastic reorganisation.
Traditional Rehabilitation: Compensation
I was always taught that we should strive for functional restitution and then use compensation strategies for what cannot be recovered. However, I believe that compensation strategies still dominate in practice, and too little effort is made to seek restitution despite what we have learned in recent years about neuroplasticity and the potential for functional rehabilitation.
"Conventional" physical and occupational therapy for chronic neurological conditions emphasises adaptive strategies: teaching patients to use assistive devices, modify environments, employ alternative movement patterns, and optimise residual function. While valuable, compensatory approaches accept permanent loss of original function and focus on maximising independence within those constraints (Evancho et al., 2023; Iddings et al., 2021).
For chronic stroke, traditional therapy after the acute window (>6 months post-stroke) historically yielded minimal upper extremity motor recovery, with patients plateauing at modest functional levels. For chronic SCI, especially motor-complete injuries, standard rehabilitation emphasised wheelchair mobility, adaptive equipment, and compensation rather than restoration of walking or hand function (Phillips et al., 2025).
Neuromodulation-Enhanced Rehabilitation: Restoration
tVNS and tSCS enable a restorative approach by temporarily creating neurobiological conditions conducive to relearning lost skills. Rather than working around deficits, combined neuromodulation-rehabilitation targets the underlying neural circuits to restore capacity (Dawson et al., 2021; Moritz et al., 2024; Korupolu et al., 2024).
A comprehensive review of combined neuromodulation and physical therapy for SCI articulates this paradigm: "Neuromodulatory stimulation approaches activate many of the same circuits that are activated by training at all levels of the neuraxis, and therefore have the potential to reverse maladaptive reorganisation associated with spinal cord injury." The review emphasises that combinatorial approaches — neuromodulation plus intensive task-specific training — are most likely to produce meaningful functional restoration (Evancho et al., 2023).
Evidence of Restorative Outcomes
The capacity for genuine restoration — recovering function thought permanently lost — is documented across both modalities:
tVNS stroke recovery: Participants in the VNS-REHAB trial averaged 2 years post-stroke (range 9 months to 10 years), well beyond the traditional window for spontaneous motor recovery. Yet 47% achieved clinically meaningful FMA-UE improvements (≥6 points), representing restored capacity for reach, grasp, and manipulation. This restoration occurred in chronic stroke survivors who had plateaued under conventional therapy (Dawson et al., 2021).
tSCS SCI recovery: Individuals with chronic motor-complete SCI (AIS A, years post-injury, no volitional movement below injury level) demonstrated improved sensation, proprioception, and voluntary activation capacity after cervical tSCS paired with training. Three participants in a chronic SCI cohort improved their ASIA Impairment Scale classifications — a remarkable outcome indicating neurological level recovery in chronic injury. Walking function improved in individuals years post-SCI who had been unable to ambulate, with gains maintained at multi-month follow-up (Inanici et al., 2020; Phillips et al., 2025; Inanici et al., 2021).
These outcomes represent restoration of function within original neural pathways (or remaining spared pathways), not merely compensation through alternative circuits.
The Timing-Dependent Restoration Mechanism
The restorative potential derives from precision timing: neuromodulator release or spinal circuit priming occurs *during* attempts at the lost function, reinforcing any residual pathway activity and progressively strengthening those connections through Hebbian plasticity.
Over weeks of paired training, functionally relevant pathways are selectively amplified while maladaptive compensatory patterns are not reinforced, gradually shifting the motor system toward pre-injury organisation patterns (Brzosko et al., 2019; Dawson et al., 2021).
This timing-dependent mechanism explains why both modalities require active patient participation and intensive training: the neuromodulation alone does not restore function; it creates windows of enhanced plasticity that must be filled with task-relevant practice to drive functional reorganisation (Evancho et al., 2023; Hays et al., 2023).
What This Means in Practice
This restorative approach appeals to patients who have plateaued under traditional therapy and are looking for something beyond adaptive equipment. It also has implications for commissioners and funders seeking interventions that may reduce long-term dependency and costs. However, a word of caution: as we work with medical devices regulated for specific clinical conditions, it is important to avoid making clinical claims beyond what the evidence and regulatory approvals support. The approach can evolve as new indications are supported by the evidence.
The critical message remains: clinical protocols must deliver on the restorative promise by integrating intensive task-specific training. The neuromodulation enables restoration, but training drives it.
Conclusion
Transcutaneous vagus nerve stimulation and transcutaneous spinal cord stimulation, despite targeting different neural levels through distinct physiological mechanisms, share fundamental principles that position both as genuinely important rehabilitation technologies. The ten parallels identified — timing-dependent pairing, session architecture, dose-response optimisation, multi-tiered carryover, plasticity priming mechanisms, parameter individualisation, home-based feasibility, exceptional safety, clinical efficacy in upper extremity rehabilitation, and restorative philosophy — create a coherent framework for understanding how modern neuromodulation fits into clinical practice.
For clinicians, the core competencies transfer directly between modalities: understanding neuromodulation as plasticity priming, synchronising stimulation with task practice, individualising parameters, and managing home transitions. For patients, the message is consistent: these are not passive treatments. They are tools that enhance what training can achieve — and they work best when paired with commitment to intensive, purposeful rehabilitation.
As rehabilitation evolves from passive interventions and adaptive strategies toward active neuromodulation paired with intensive training, the parallels between tVNS and tSCS provide a roadmap for evidence-based implementation. Both modalities represent the direction in which neurological rehabilitation is heading — away from compensation, toward genuine functional restoration through neuroplastic reorganisation.
References
Badran BW, Huffman SM, Deng ZD, et al. A pilot randomized controlled trial of supervised, at-home, self-administered transcutaneous auricular vagus nerve stimulation (taVNS) to manage long COVID symptoms. Bioelectronic Medicine. 2022;8:13. DOI: 10.1186/s42234-022-00094-y. PMID: 35765105.
Badran BW, Jenkins DD, DeVries WH, et al. Remotely supervised at-home delivery of taVNS for autism spectrum disorder: feasibility and preliminary efficacy. Frontiers in Psychiatry. 2023;14:1238328. DOI: 10.3389/fpsyt.2023.1238328.
Baig SS, Kamarova M, Ali A, et al. Transcutaneous vagus nerve stimulation (tVNS) in stroke: the evidence, challenges and future directions. Autonomic Neuroscience. 2022;237:102909. DOI: 10.1016/j.autneu.2021.102909. PMID: 34861612.
Brzosko Z, Mierau SB, Paulsen O. Neuromodulation of spike-timing-dependent plasticity: past, present, and future. Neuron. 2019;103(4):563-581. DOI: 10.1016/j.neuron.2019.05.041. PMID: 31437453.
Butt MF, Albusoda A, Farmer AD, Aziz Q. The anatomical basis for transcutaneous auricular vagus nerve stimulation. Journal of Anatomy. 2020;236(4):588-611. DOI: 10.1111/joa.13122. PMID: 31742681.
D'Agostini M, Chrousos A, Nijs M, et al. Transcutaneous vagus nerve stimulation boosts post-error accuracy in perceptual decision-making. Brain Stimulation. 2024. DOI: 10.1016/j.brs.2024.10.021.
Dawson J, Pierce D, Dixit A, et al. Vagus nerve stimulation paired with rehabilitation for upper limb motor function after ischaemic stroke (VNS-REHAB): a randomised, blinded, pivotal, device trial. The Lancet. 2021;397(10284):1545-1553. DOI: 10.1016/S0140-6736(21)00475-X. PMID: 33894832.
Deer TR, Pope JE, Lamer TJ, et al. The Predictive Value of Transcutaneous Electrical Nerve Stimulation (TENS) as a predictor of peripheral nerve stimulation response. Neuromodulation. 2020;23(7):920-926. DOI: 10.1111/ner.13211. PMID: 32757257.
Englot DJ, Rolston JD, Wright CW, et al. Rates and predictors of seizure freedom with vagus nerve stimulation for intractable epilepsy. Neurosurgery. 2016;79(3):345-353. DOI: 10.1227/NEU.0000000000001165. PMID: 26645965.
Evancho A, Tyler WJ, McGregor K. A review of combined neuromodulation and physical therapy interventions for enhanced neurorehabilitation. Frontiers in Human Neuroscience. 2023;17:1151218. DOI: 10.3389/fnhum.2023.1151218.
Farmer AD, Strzelczyk A, Finisguerra A, et al. International consensus-based review of the mechanisms, indications, and perspectives of transcutaneous vagus nerve stimulation. European Journal of Neurology. 2021;28(10):3211-3224. DOI: 10.1111/ene.14982.
Feldman DE. The spike-timing dependence of plasticity. Neuron. 2012;75(4):556-571. DOI: 10.1016/j.neuron.2012.08.001. PMID: 22920249.
Gilligan C, Volschenk W, Russo M, et al. Clinical predictors of pain relief with 60-day peripheral nerve stimulation. Pain Practice. 2023;23(8):869-876. DOI: 10.1111/papr.13267.
Hays SA. Enhancing rehabilitative therapies with vagus nerve stimulation. Neurotherapeutics. 2016;13(2):382-394. DOI: 10.1007/s13311-015-0417-z. PMID: 26671658.
Hays SA, Rennaker RL, Kilgard MP. How to fail with paired VNS therapy. Brain Stimulation. 2023;16(5):1252-1258. DOI: 10.1016/j.brs.2023.08.009. PMID: 37595833.
Hofstoetter US, Freundl B, Binder H, Minassian K. Transcutaneous spinal cord stimulation enhances walking performance and reduces spasticity in individuals with multiple sclerosis. Brain Sciences. 2021;11(4):472. DOI: 10.3390/brainsci11040472. PMID: 33917523.
Hofstoetter US, Freundl B, Danner SM, et al. Transcutaneous spinal cord stimulation induces temporary attenuation of spasticity in individuals with spinal cord injury. Journal of Neurotrauma. 2020;37(3):481-493. DOI: 10.1089/neu.2019.6588. PMID: 31333064.
Hofstoetter US, Laliberte AM, Bhatt DK, et al. Transcutaneous spinal cord stimulation neuromodulates presynaptic and postsynaptic inhibition to reduce spasticity after spinal cord injury. Journal of Neurophysiology. 2024;132(6):1774-1789. DOI: 10.1152/jn.00274.2024.
Iddings JA, Zarkou A, Field-Fote EC. Noninvasive neuromodulation and rehabilitation to promote functional restoration in persons with spinal cord injury. Current Opinion in Neurology. 2021;34(6):812-818. DOI: 10.1097/WCO.0000000000000997. PMID: 34766554.
Inanici F, Brighton LN, Samejima S, et al. Transcutaneous spinal cord stimulation promotes long-term recovery of upper extremity function in chronic tetraplegia. IEEE Transactions on Neural Systems and Rehabilitation Engineering. 2020;28(6):1312-1320. DOI: 10.1109/TNSRE.2020.2984960. PMID: 32248117.
Inanici F, Samejima S, Gad P, et al. Targeted transcutaneous spinal cord stimulation promotes persistent recovery of upper extremity strength and tactile sensation in spinal cord injury: a pilot study. Journal of Clinical Medicine. 2021;10(18):4141. DOI: 10.3390/jcm10184141.
Kampusch S, Kaniusas E, Thurnher MM, et al. Safety of transcutaneous auricular vagus nerve stimulation (taVNS): a systematic review and meta-analysis. Scientific Reports. 2022;12:22055. DOI: 10.1038/s41598-022-22055-9. PMID: 36543829.
Korupolu R, Miller A, Park A, Yozbatiran N. Neurorehabilitation with vagus nerve stimulation: a systematic review. Frontiers in Neurology. 2024;15:1390217. DOI: 10.3389/fneur.2024.1390217. PMID: 38872818.
Malloy DC, Cote M-P. Multi-session transcutaneous spinal cord stimulation prevents chloride homeostasis imbalance and the development of hyperreflexia after spinal cord injury in rat. Experimental Neurology. 2024;376:114754. DOI: 10.1016/j.expneurol.2024.114754. PMID: 38493983.
Malloy DC, Cote M-P. Transcutaneous spinal cord stimulation at alternating intensities preferentially prevents the development of spasticity after contusion SCI in rat. The Journal of Physiology. 2025;603(17):4845-4865. DOI: 10.1113/JP287100.
Meier K, de Vos CC, Bordeleau M, et al. Examining the duration of carryover effect in patients with chronic pain treated with spinal cord stimulation (EChO study). Neuromodulation. 2024;27(5):887-898. DOI: 10.1016/j.neurom.2024.01.002. PMID: 38456888.
Moritz C, Field-Fote EC, Tefertiller C, et al. Non-invasive spinal cord electrical stimulation for arm and hand function in chronic tetraplegia: a safety and efficacy trial. Nature Medicine. 2024;30(6):1276-1283. DOI: 10.1038/s41591-024-02940-9. PMID: 38769431.
Phillips AA, Squair JW, Sayenko DG, et al. Safety and effectiveness of multisite transcutaneous spinal cord stimulation combined with activity‑based therapy when delivered in a community rehabilitation setting: a real‑world pilot study. Neuromodulation. 2025. (Ahead of print / online first; ClinicalTrials.gov NCT04132596).
Pruitt DT, Hakim S, Thompson A, et al. Optimizing dosing of vagus nerve stimulation for stroke recovery. Brain Stimulation. 2020;13(6):1573-1581. DOI: 10.1016/j.brs.2020.06.010. PMID: 32583333.
Redgrave J, Day D, Leung H, et al. Safety and tolerability of Transcutaneous Vagus Nerve Stimulation in humans; a systematic review. Brain Stimulation. 2018;11(6):1225-1238. DOI: 10.1016/j.brs.2018.08.010. PMID: 30217648.
Ruiz AD, Malley KM, Danaphongse TT, et al. Effective delivery of vagus nerve stimulation requires many stimulations per session and many sessions per week over many weeks to improve recovery of somatosensation. Neurorehabilitation and Neural Repair. 2023;37(9):652-661. DOI: 10.1177/15459683231197412. PMID: 37694568.
Strawn JR, Goldschmied JR, Zhaoyang R, et al. Optimizing transcutaneous vagus nerve stimulation parameters for sleep and autonomic function in veterans with posttraumatic stress disorder with or without mild traumatic brain injury. Sleep. 2025;48(8):zsaf152. doi:10.1093/sleep/zsaf152.
Svensson TH, Bundy DT, Chen X, et al. Cervical transcutaneous vagal nerve stimulation (ctVNS) improves objective measures of arousal and multitasking. Communications Biology. 2021;4:735. DOI: 10.1038/s42003-021-02145-7.
Tefertiller C, Moritz C, Field-Fote EC, et al. Non-invasive transcutaneous spinal cord stimulation for upper extremity rehabilitation in chronic tetraplegia: the Up-LIFT framework. Archives of Physical Medicine and Rehabilitation. 2024;105(10):1870-1879. DOI: 10.1016/j.apmr.2024.06.004. PMID: 38958629.
Tefertiller C, Trumbower RD, Morse LR, et al. Home-based noninvasive spinal cord stimulation safely enhances hand and arm function in people with spinal cord injury. Neurology: Clinical Practice. 2025;15(6):e200537. doi:10.1212/CPJ.0000000000200537. Epub 2025 Sep 24.
Yaghouby F, et al. Critical review of transcutaneous vagus nerve stimulation: challenges for translation to clinical practice. Bioelectronic Medicine. 2024;6:4. DOI: 10.1186/s42234-020-00044-6.