Cauda Equina Syndrome and Denervated Muscle: Your Options for Long-Term Health
Cauda equina syndrome (CES) is one of the most challenging situations in spinal cord injury rehabilitation. Unlike injuries higher in the spine, CES directly damages the lower motor neurons—the nerve cells that connect to and control the leg muscles. This results in the muscles becoming denervated, losing their nerve supply completely.
For many years, people with CES were told little could be done about the muscle wasting that occurs. The common belief was that denervated muscles would inevitably weaken, and electrical stimulation—which is effective for higher spinal injuries—simply wouldn't help. That perspective has shifted.
Research over the past twenty years shows that denervated muscles can be preserved and even improved with appropriate electrical stimulation—however, it requires a different approach from standard rehabilitation methods. In this article, I will explain what happens to muscles after cauda equina syndrome, why conventional methods often fail, and what options are available for maintaining long-term muscle and tissue health.
Understanding Cauda Equina Syndrome
The cauda equina (Latin for "horse's tail") is the bundle of nerve roots that extends below the end of the spinal cord, typically from around the L1-L2 vertebral level downward. These nerve roots carry signals to and from the lower limbs, bladder, bowel, and sexual organs.
Cauda equina syndrome occurs when these nerve roots are compressed or damaged. The most common cause is a large disc herniation compressing the nerve roots, but CES can also result from spinal stenosis (narrowing of the spinal canal), trauma such as fractures or dislocations, tumours, infection, or rarely as a complication of spinal surgery.
CES is a medical emergency. The sooner surgical decompression occurs, the better the chance of preserving nerve function. A meta-analysis by Ahn and colleagues found that surgery performed within 48 hours consistently yields better outcomes, whereas delays beyond 24 hours significantly increase the relative risk of poor urinary outcomes (Ahn et al., 2000, Spine). However, even with prompt treatment, many people are left with permanent deficits.
How Common Is Cauda Equina Syndrome?
Until recently, CES was thought to be rare—historical estimates suggested fewer than 1 case per 100,000 people annually. However, a prospective UK-wide cohort study published in 2022 found the incidence to be considerably higher. Woodfield and colleagues, studying the Scottish population, identified an incidence of 2.7 per 100,000 (95% CI: 2.3-3.2)—at least four times higher than previous European estimates (Woodfield et al., 2022, Spine). The incidence was particularly high among women aged 30-39, reaching 7.2 per 100,000.
In England, approximately 8,000 suspected cases are reported annually, though only about 19% are radiologically and clinically confirmed (Hoeritzauer et al., 2020, JAMA Network Open). These figures show that if you have CES, you are far from alone—and that more people live with the long-term consequences than was previously recognised.
Long-Term Outcomes After Cauda Equina Syndrome
Understanding the long-term course of CES helps explain why proactive intervention is crucial. Long-term follow-up studies present a sobering view of ongoing dysfunction that extends far beyond the initial acute phase.
A prospective study by Katzouraki and colleagues tracked patients for an average of 43 months after CES and found that 76% still experienced bladder dysfunction—much higher than earlier studies reporting 41-47%, likely due to the use of more sensitive assessment tools (Katzouraki et al., 2019, European Spine Journal). Bowel dysfunction affected 13% of patients, while 39% reported sexual dysfunction, with significantly worse outcomes in those who had progressed to CES with urinary retention (CES-R) compared to incomplete CES (CES-I).
Perhaps most relevant to this discussion, 76% of patients reported motor weakness at follow-up—new weakness that had not resolved since surgery. Physical dysfunction, including persistent weakness, sensory loss, or pain, impacted 48% of patients. Further research at 3-4 year follow-up revealed additional consequences: 90% reported serious bladder problems, 64% experienced worsened low back pain, 68% developed neuropathic leg pain, and 32% suffered from neuropathic pelvic pain.
Fatigue affected 94%, and 78% developed depression (Srikandarajah et al., 2024, *Bulletin of the Royal College of Surgeons of England*). These figures highlight that CES is not merely an acute emergency to be managed and forgotten. It is a condition that deeply influences long-term quality of life, making it vital to maintain muscle and tissue health for those impacted.
Why CES Is Different From Higher Spinal Cord Injuries
Understanding the differences between upper motor neuron (UMN) and lower motor neuron (LMN) injuries is essential for understanding why CES requires a different rehabilitation approach.
Upper motor neuron injuries, such as spinal cord injuries at the thoracic level T11 and above, damage the pathways from the brain but leave the lower motor neurons intact. The nerves that connect directly to muscles are still present and functional—they just cannot receive signals from the brain. This is why people with injuries higher in the spinal cord often have spastic (tight, reflexive) muscles rather than flaccid ones.
Lower motor neuron injuries, including CES, conus medullaris syndrome, and injuries at T12 and below, damage the motor neurons themselves. The nerve cells that directly control muscle fibres are destroyed or severely impaired. Without these neurons, muscles become denervated—disconnected from any nerve supply.
KEY POINT: In CES, the muscles lose their nerve connection entirely. This is fundamentally different from higher spinal injuries where nerves remain intact but are disconnected from brain control. This difference has significant implications for treatment.
With an upper motor neuron injury, muscle tone is often spastic (tight). Reflexes are present or exaggerated. Muscle wasting is slow and less severe. Standard NMES and FES are effective, and denervated-muscle stimulation is not needed.
With a lower motor neuron injury, as with CES, muscle tone is flaccid, reflexes are absent or diminished, and muscle wasting is rapid and severe. Standard NMES and FES do not work, and de-inhibited muscle stimulation is required.
The Biology of Muscle Denervation: What Happens at the Cellular Level
When a muscle loses its nerve supply, a cascade of molecular and cellular changes begins that explains both the dramatic atrophy that follows and why the timing of intervention matters so much.
The Protein Degradation Machinery
Two major protein degradation systems become hyperactive in denervated muscle. The ubiquitin-proteasome system (UPS) relies on two muscle-specific enzymes called E3 ubiquitin ligases—MuRF-1 and Atrogin-1 (also known as MAFbx)—which become significantly upregulated following denervation. These enzymes effectively tag the structural proteins that give muscle its bulk and strength for destruction. Simultaneously, the autophagy-lysosome system activates, further accelerating the breakdown of long-lived proteins and cellular components (Bonaldo & Sandri, 2013, Disease Models & Mechanisms).
The signalling pathways driving this process have been mapped in considerable detail. Under normal circumstances, the FoxO3 transcription factors are suppressed by a pathway involving the protein Akt1. Denervation removes this suppression, allowing FoxO3 to drive expression of the very genes (MAFbx, MuRF1, and others) that cause protein breakdown. Myostatin, one of the most powerful atrophy-inducing molecules known, also increases its activity through the Smad2/3 pathway. Additional pathways involving NF-kappaB and histone deacetylases (HDAC4/HDAC5) contribute to a coordinated programme of muscle destruction (Shen et al., 2020, Annals of Translational Medicine).
Transcriptome analysis of denervated muscle at 14 days post-injury reveals the scale of these changes: 2,749 transcripts are upregulated (primarily involved in protein binding and degradation) while 2,941 transcripts are downregulated, including genes essential for energy metabolism such as those involved in oxidative phosphorylation, the citric acid cycle, and glycolysis (Ji et al., 2019, Frontiers in Physiology).
Why Fat and Fibrosis Replace Muscle Tissue
The replacement of muscle by fat and fibrous tissue is not simply passive—it is actively driven by specific molecular signals. TGF-beta signalling pathway activation plays a critical role in promoting fibrosis, becoming particularly evident after about 14 days post-denervation. The proliferating connective tissue that encircles denervated muscle fibres may actually prevent infiltration of regenerating axons, creating a barrier to recovery even if nerve regrowth were to occur (Carlson, 2014, European Journal of Translational Myology).
The Puzzle of Satellite Cells
One might expect that muscle stem cells—the satellite cells that normally regenerate muscle after injury—could rescue the situation. The reality is more complex. Human satellite cells can survive denervation for remarkably long periods; cells isolated from muscle denervated for over 20 years retain the ability to engraft and form new muscle fibre when transplanted into suitable environments (Schaaf et al., 2020, *Stem Cells*).
However, within the denervated muscle itself, regeneration becomes abortive. At 10 days post-denervation, satellite cell frequency increases to approximately 11% (compared to 6% in normal muscle), indicating activation. Yet this activation does not translate to effective regeneration. Myogenesis initiated in denervated muscle does not lead to the formation of normal muscle fibres, despite the presence of activated satellite cells. A deficit in regulatory factors controlling satellite cell activity appears responsible for this failure (Borisov et al., 2005, Anatomy and Embryology).
Changes to Muscle Fibre Types
Denervation causes complex changes to muscle fibre composition. Initially, expression of slow myosin heavy chain (MyHC) rises from 68% to 88% within 3 weeks and approaches 100% by 7 weeks. However, this increase occurs without a parallel increase in adult/mature slow MyHC—the fibres develop mixed fast-plus-slow profiles rather than becoming true slow fibres. Regional differences exist, with changes being greater in proximal portions of muscles.
After more than 6 months of permanent denervation, a remarkable transformation occurs: muscles undergo almost complete transformation into nearly pure fast muscles (Patterson et al., 2006, Journal of Applied Physiology). Understanding these fibre type shifts has implications for stimulation protocols, which may need to adapt to the changing properties of the muscle over time.
The Timeline of Deterioration
When muscles lose their nerve supply, a predictable sequence of deterioration begins that can be tracked through imaging and histology.
In the early phase (weeks to months), muscle fibres begin to atrophy, the muscle loses its ability to contract voluntarily, standard electrical stimulation produces no response, and muscle mass visibly decreases. On MRI, increased signal is seen on STIR/T2 sequences, reflecting oedema, while T1-weighted images remain normal.
During the intermediate phase (months to years), continued loss of muscle bulk occurs alongside progressive replacement of muscle tissue with fat and fibrous tissue. Changes to the muscle's internal structure include loss of organised myofibrils, and reduced blood supply to the affected areas becomes evident. T1 signal on MRI begins to increase, reflecting early fat infiltration.
In the late phase (years), severe atrophy develops—muscles may be barely recognisable, with muscle wet weight decreasing to just 10-20% of healthy muscle weight. Extensive fat infiltration occurs, fibrosis (scarring) of muscle tissue develops, and there is a significantly increased risk of pressure ulcers due to loss of tissue padding alongside impaired circulation in the lower limbs.
Is There a "Point of No Return"?
The research suggests there is indeed a critical window, though it is not absolute. Cross-sectional area and adipose fraction density continue to worsen through at least 2 months post-injury. By 6-12 months, fat infiltration becomes clearly visible on T1 MRI, and structural changes become increasingly difficult to reverse. The literature suggests that after approximately one year, changes are considered largely irreversible on imaging. By 2-3 years, severe atrophy develops with fibre size decreasing to just 10% of normal (May et al., 2000, American Journal of Roentgenology; Carlson, 2014).
This timeline creates genuine urgency—but as we shall see, the situation is not hopeless even for those who discover these options years after injury.
Why Standard Electrical Stimulation Doesn't Work: The Electrophysiology
If you've tried standard FES (Functional Electrical Stimulation) or NMES (Neuromuscular Electrical Stimulation) devices and found they produce no response in your legs, you've discovered the fundamental limitation of these technologies for denervated muscle.
Standard FES and NMES work by stimulating motor nerves. The electrical pulse triggers the nerve, which then activates the muscle. This works brilliantly for people with upper motor neuron injuries because their motor nerves are intact—the devices simply provide the activation signal that the brain can no longer deliver.
With CES, those motor nerves are damaged or absent. There's nothing for the electrical pulse to stimulate. The signal has nowhere to go.
Significantly different forms of electrical stimulation are necessary to work with denervated muscles.
The Membrane Changes That Explain Everything
The reason denervated muscle requires such different stimulation parameters lies in fundamental changes to the electrical properties of the muscle fibre membranes themselves. Two key measurements capture this: rheobase (the minimum current strength required to excite the muscle given unlimited time) and chronaxie (the duration of stimulus required when using twice the rheobase current).
In an innervated muscle, chronaxie is typically less than 1 millisecond. In denervated muscle, chronaxie increases dramatically, to between 9.5 and 30 milliseconds, a 10- to 30-fold increase. Longitudinal studies using implanted stimulators in animal models found that chronaxie increased 3-fold from 4.5 ms to 14.1 ms during the first two weeks post-denervation and remained elevated throughout the experimental period (Ashley et al., 2005, Artificial Organs).
This increase in chronaxie is directly related to changes in the density of voltage-gated sodium channels in the cell membrane. Denervation reduces this density, which directly affects excitability. Since chronaxie is inversely proportional to excitability, the greater chronaxie values indicate that denervated fibres require electrical current to persist for substantially longer periods before the excitation process can be initiated.
Additionally, the resting membrane potential of denervated muscle fibres depolarises (becomes less negative). Normal innervated muscle maintains a resting potential of approximately -82 to -90 mV. After denervation, this falls to approximately -68 mV within just 2 days. This depolarisation occurs rapidly near the nerve section site and reflects impaired Na+/K+-ATPase pump activity, leading to reduced intracellular potassium and elevated intracellular sodium (McArdle, 1983, Progress in Neurobiology).
These fundamental changes in membrane properties explain precisely why standard stimulators with pulse widths of 0.1-0.5 milliseconds cannot activate denervated muscle—the pulses are simply too brief to trigger depolarisation in fibres with chronaxie values that are dramatically elevated.
The Alternative: Direct Muscle Fibre Stimulation
The good news is that muscle fibres themselves can be made to contract—even without any nerve supply—if you deliver electrical energy directly to them. This requires a fundamentally different stimulation approach.
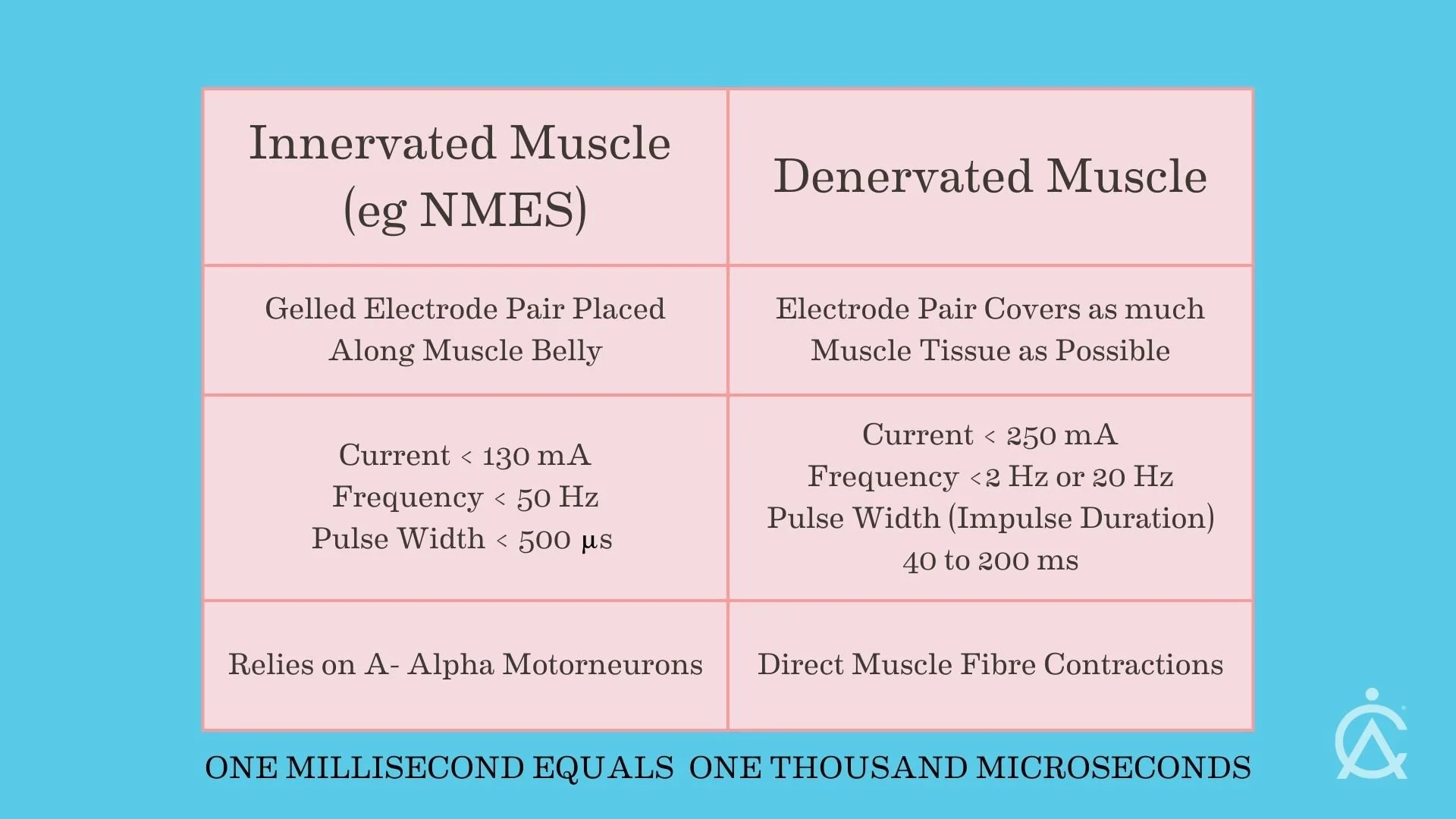
The key differences are evident in the stimulation parameters. The most commonly used waveforms are so-called biphasic and typically are expressed with the following parameters:
For NMES and FES, pulse widths typically range from 0.1 to 0.5 ms. Current values are up to 130 mA. The applied frequency is between 20 and 50 Hz, and electrodes can be gel pads.
For denervated muscle stimulation, pulse widths are typically between 100 and 200 ms, and sometimes even longer. Current values can be up to 250 mA in exceptional cases. Frequencies depend on the stage of rehabilitation, ranging from 0.5 to 5 Hz in the early stages of training and 20 to 30 Hz in later stages. We often use large, wet sponge carbon-rubber electrodes to cover as much of the muscle tissue as possible. If gel-type electrodes are used, great care must be taken due to the potentially high current densities.
The dramatically longer pulse widths (100-400 times those of standard devices) are necessary because of the elevated chronaxie values we discussed. The membrane properties of denervated muscle fibres mean they are harder to excite. Only with a sufficiently long pulse duration can electrical energy directly depolarise and contract the fibre.
The Evidence: What Can Denervated Muscle Stimulation Actually Achieve?
The most substantial evidence base comes from the European RISE project (Rescue of denervated and degenerated muscle), led by Professor Helmut Kern in Vienna. This multinational research programme demonstrated that home-based functional electrical stimulation with appropriate parameters could produce remarkable improvements in permanently denervated muscles.
Specific Outcomes from the Research
The RISE project conducted a 2-year longitudinal prospective study of 25 patients with complete conus/cauda equina lesions using home-based functional electrical stimulation (h-bFES). The outcomes were measured with rigorous methodology including CT imaging, muscle biopsies, and force measurements.
Muscle cross-sectional area: CT imaging documented a mean quadriceps increase of 29.74% (range 10.45% to 58%) after approximately one year of treatment (Kern et al., 2005, Artificial Organs).
Muscle fibre diameter: Histological analysis of biopsies showed an average increase of 59% in muscle fibre diameter after 8 months of FES training (Kern & Carraro, 2014, Basic Applied Myology).
Force production: Perhaps most striking was the 1,187% increase in force output during electrical stimulation. Quadriceps torque increased from 0.8 ± 1.3 to 10.3 ± 8.1 N.m (P<0.001). Remarkably, 25% of subjects became able to perform FES-assisted stand-up exercises (Kern et al., 2010, Neurorehabilitation and Neural Repair).
What Biopsies Reveal
Muscle biopsies provided direct evidence of structural improvement. Before stimulation, samples showed ultrastructural disorganisation of myofibrils, myofibril-free zones, severe atrophy with nuclear clumping, and fibro-fatty degeneration. After sustained stimulation, biopsies demonstrated improvements in the ultrastructural organisation of contractile material, reductions in fat and connective tissue within the muscle, increases in both aerobic and anaerobic muscle enzymes to within the normal range, and fibre hypertrophy, with intracellular structural benefits occurring even without axonal neurotrophic substances.
Particularly intriguing was evidence of new fibre regeneration (hyperplasia), not just hypertrophy of existing fibres (Kern et al., 2004, Artificial Organ).
Vascular Changes
Echo Doppler analysis revealed that electrical stimulation induced low-resistance arterial flow with hyperaemia lasting longer than the stimulation duration itself, suggesting a mechanism supporting the trophic effects of treatment beyond the mechanical benefits of contraction.
Realistic Expectations for CES
Let me be clear about realistic expectations. With complete, permanent denervation—as typically occurs with CES—the primary goals are tissue preservation and long-term health rather than functional recovery.
Realistic goals include preserving existing muscle mass and preventing further atrophy, maintaining or improving tissue quality by reducing fat infiltration and fibrosis, improving local circulation in the lower limbs, reducing the risk of pressure ulcers by maintaining tissue padding, preserving the contractile capability of muscle fibres, and maintaining body composition and metabolic health.
Less realistic goals include restoring voluntary movement (nerve damage prevents this), achieving independent walking (though some research participants achieved assisted standing), and expecting rapid or dramatic, visible changes (improvements are gradual).
That said, the tissue preservation goals are genuinely valuable. Preventing pressure ulcers alone can make an enormous difference to quality of life—anyone who has experienced a serious pressure sore knows how debilitating and slow to heal they can be.
KEY POINT: For CES, the primary benefit of denervated muscle stimulation is long-term tissue health—preventing complications rather than restoring function. This is still highly valuable.
Which Muscles Can Be Treated?
In CES, the muscles affected depend on which nerve roots are damaged. Commonly affected muscles include the quadriceps (front of thigh), which are the primary target in most denervated muscle programmes. They are large, accessible, and respond well to stimulation. The research literature focuses heavily on the quadriceps, and the protocols are well-established.
The hamstrings (back of the thigh) benefit both from direct stimulation and from the mechanical stretching that occurs when the quadriceps contract. Research shows hamstring improvements even when they are not directly targeted, though direct stimulation can also be applied.
The gluteal muscles (buttocks) are important for sitting tolerance and pressure distribution. Stimulating the gluteals requires careful positioning—some people use standing frames to achieve appropriate access. The gluteals are particularly important for preventing pressure ulcers.
The calf muscles can be targeted, though they may require separate electrode setups.
The muscles of bladder and bowel control are also affected by CES but are not amenable to this type of external electrical stimulation.
The Practical Reality: What's Involved
If you're considering denervated muscle stimulation for CES, here's what's actually involved.
Equipment includes a stimulator capable of producing the necessary parameters (such as the [RISE Stimulator](/rise-stimulator-electrotherapy-device)), appropriate electrodes—typically large wet sponge electrodes with carbon rubber backing, and positioning equipment (cushions, straps) to maintain electrode contact.
Time commitment involves 30 minutes per muscle group per session, 5-6 sessions per week, and an ongoing commitment measured in months and years.
The protocol progression typically moves through three phases. In the twitch phase, long, low-frequency pulses produce individual muscle twitches, restoring basic muscle excitability. As the muscle becomes more responsive, parameters are adjusted in the tetanic phase to produce sustained (tetanic) contractions, which build strength and mass more effectively. Once muscle condition is optimised, the frequency can be reduced in a maintenance phase to preserve gains.
Support required includes an initial assessment and training from someone experienced in denervated muscle treatment, periodic reviews (typically every 6 months) to assess progress and adjust protocols, and ongoing access to guidance for troubleshooting.
Challenges specific to CES include reduced or absent sensation, requiring visual confirmation of contractions rather than feeling them; awkward positioning for gluteal stimulation; and a substantial, ongoing time commitment.
When to Start: The Evidence on Timing
The research is clear: earlier is better. The structural integrity of muscle fibres degrades over time following denervation, and the cellular and molecular changes discussed earlier narrow the window of opportunity.
The optimal intervention window appears to be 20 days to 3-6 months post-injury, based on research showing that repairs at 20 days result in better functional recovery than immediate intervention (suggesting that some natural healing processes benefit from a brief delay), but this advantage diminishes as delays extend beyond this window. After 6-12 months, structural changes become increasingly irreversible (Carlson, 2014).
Imaging studies track this decline clearly. In the days to weeks after injury, MRI shows increased signal on STIR/T2 sequences (oedema) with normal T1. Over weeks to months, reduced bulk becomes apparent and the T1 signal increases (early fat). After approximately one year, changes are considered largely irreversible on imaging. By 2-3 years, severe atrophy and extensive fatty infiltration are evident on CT (May et al., 2000).
What About Starting Years After Injury?
If you've only recently learned about this option years after your CES, don't assume it's too late. The RISE project specifically investigated outcomes in patients who began treatment long after injury.
Encouraging findings from delayed intervention show that h-bFES, initiated as late as 3 years post-SCI, can recover hamstring muscles, with post-training analyses performed after 5 years of permanent muscle denervation. A case study of h-bFES applied 26 months post-injury showed increased muscle excitability, size, force capacity, and induced knee extension. Histology in late starters showed reduced fat and connective tissue, myofibre hypertrophy, and evidence of new fibre regeneration (hyperplasia) (Kern et al., 2016, European Journal of Translational Myology).
However, the extent of recovery decreases with increasing years of denervation. Many historical interventions were initiated more than 8 months post-SCI because of prior beliefs that stimulation was ineffective, whereas better outcomes are seen within 1-2 years. Even so, trials are still recommended even in long-term denervation, given the low risk profile (Carraro et al., 2020, European Journal of Translational Myology).
KEY POINT: If you have CES, enquire about denervated muscle stimulation sooner rather than later. Early intervention produces better outcomes—but even late intervention can produce meaningful improvements.
A Common Question: Does Stimulation Affect Reinnervation?
In cases where some recovery of nerve function is possible—particularly with incomplete CES—people understandably worry whether electrical stimulation might interfere with the natural process of reinnervation. This concern has been studied directly.
The evidence is generally reassuring. In studies of denervated canine muscle, chronic stimulation actually increased correct reinnervation (P<0.0064), reduced incorrect reinnervation (P<0.0084), and showed a trend toward enhancing overall reinnervation magnitude (Zealear et al., 2002, *Laryngoscope*). In rat studies, stimulation post-crush injury preserved muscle mass and tetanic strength without inhibiting recovery. Kern and colleagues' studies on denervated human muscles found that FES reversed degeneration without evidence of inhibiting regeneration.
Some animal studies have suggested that certain stimulation protocols may delay reinnervation, particularly when daily stimulation is used for 6-14 days post-denervation in rat models. The effects appear to vary with protocol intensity, timing, muscle type, and species. However, the clinical consensus, particularly based on the Vienna group's extensive experience, is that stimulation does not inhibit reinnervation and may even facilitate it (Kern et al., 2010).
The practical implication is significant. Given that stimulation does not appear to inhibit reinnervation, that the window for muscle preservation is limited, and that delay risks irreversible muscle loss, starting stimulation early while monitoring for signs of reinnervation appears to be the evidence-supported approach.
Serial electromyography (EMG) can help monitor for signs of reinnervation. Fibrillation potentials and positive sharp waves indicate ongoing denervation, while nascent motor unit potentials (NMUPs)—small-amplitude, short-duration, polyphasic potentials—indicate early axonal reinnervation. These signals can appear before clinical improvement is evident, providing guidance for protocol adjustment (Dumitru & Amato, 2002, Muscle & Nerve).
What Success Looks Like
For someone with CES using denervated muscle stimulation appropriately, success might look like maintaining thigh circumference measurements rather than continuing to lose mass, achieving consistent, visible muscle contractions during stimulation sessions, improved tissue firmness on examination, no development of pressure ulcers over the years of use, stable sitting tolerance and comfort, and on imaging (if performed), preservation of muscle tissue rather than progressive fat replacement. Put simply, electrical stimulation can restore tissue bulk and quality of muscle tissue.
These may seem modest compared to the dramatic recovery stories sometimes associated with rehabilitation technology. But for someone living with CES, preventing the complications of progressive muscle loss represents a genuine and meaningful improvement in long-term health and quality of life.
Getting Started
If you have cauda equina syndrome and are interested in exploring denervated muscle stimulation, the first step is proper assessment. We need to understand the extent and completeness of your denervation, which muscles are affected, how long since your injury, your current muscle status (ideally with baseline measurements), your ability to commit to the required protocol, and any other health factors that might influence treatment.
At Anatomical Concepts, we work with people throughout the UK who have CES and other lower motor neuron injuries. We provide assessment, equipment setup and training, and ongoing support.
If you'd like to discuss whether denervated muscle stimulation might be appropriate for your situation, please [contact us](/contact). We're happy to have an initial conversation to help you understand your options.
References
Ahn UM, et al. (2000). "Cauda equina syndrome secondary to lumbar disc herniation: a meta-analysis of surgical outcomes." *Spine*. PMID: 18377315
Ashley Z, et al. (2005). "Determination of the chronaxie and rheobase of denervated limb muscles in conscious rabbits." *Artificial Organs*. 29(3). PMID: 15725219
Bonaldo P, Sandri M. (2013). "Cellular and molecular mechanisms of muscle atrophy." *Disease Models & Mechanisms*. 6(1):25-39. PMC3424188
Borisov AB, et al. (2005). "Abortive myogenesis in denervated skeletal muscle: differentiative properties of satellite cells, their migration, and block of terminal differentiation." *Anatomy and Embryology*. 209(4):269-279. PMID: 15761724
Carlson BM. (2014). "The biology of long-term denervated skeletal muscle." *European Journal of Translational Myology*. 24(1):5-11. PMC4239769
Carraro U, et al. (2020). "Recovery of denervated muscles by functional electrical stimulation." *European Journal of Translational Myology*. PMC7460102
Dumitru D, Amato AA, Zwarts MJ (eds.). (2002). Electrodiagnostic Medicine. 2nd ed. Philadelphia: Hanley & Belfus.
Hoeritzauer I, et al. (2020). "What is the incidence of cauda equina syndrome? A systematic review." *JAMA Network Open*. PMID: 32059184
Ji Y, et al. (2019). "Investigation of the time course of denervation-induced skeletal muscle atrophy." *Frontiers in Physiology*. 10:1298
Katzouraki G, et al. (2019). "A prospective study of the long-term outcome of patients with cauda equina syndrome." *European Spine Journal*. PMC6704093
Kern H, et al. (2004). "Denervated muscles in humans: limitations and problems of currently used functional electrical stimulation training protocols." *Artificial Organs*. PMID: 15025217
Kern H, et al. (2005). "Electrical stimulation of denervated muscles: first results of a clinical study." *Artificial Organs*. 29(3):203-206
Kern H, et al. (2010). "Functional electrical stimulation of long-term denervated muscles in humans." *Neurorehabilitation and Neural Repair*. 24(8):709-21. PMID: 20460493
Kern H, Carraro U. (2014). "Home-based functional electrical stimulation of human permanent denervated muscles." *Basic Applied Myology*. 24(1):1-27
Kern H, et al. (2016). "Home-based functional electrical stimulation rescues permanently denervated muscles in paraplegic patients." *European Journal of Translational Myology*. PMC4749003
May DA, et al. (2000). "MR imaging signs of peripheral nerve injury and denervation." *American Journal of Roentgenology*. 175:1099-1104
McArdle JJ. (1983). "Molecular aspects of the trophic influence of nerve on muscle." *Progress in Neurobiology*. PMC1309272
Patterson MF, et al. (2006). "Denervation produces different single fiber phenotype changes in fast- and slow-twitch hindlimb muscles of the rat." *Journal of Applied Physiology*. PMC3008217
Schaaf GJ, et al. (2020). "Human satellite cells from muscle denervated for extended periods retain regenerative capacity." *Stem Cells*. Wiley
Shen Y, et al. (2020). "Transcriptome signature of denervation-induced skeletal muscle atrophy." *Annals of Translational Medicine*. ATM-68455
Srikandarajah N et al. Cauda Equina Syndrome Core Outcome Set (CESCOS). PLoS One. 2020;15(1):e0225907.
Woodfield J, et al. (2022). "Incidence of cauda equina syndrome: a prospective UK-wide cohort study." *Spine*. PMID: 36315989
Zealear DL, et al. (2002). "Reinnervation of the denervated canine posterior cricoarytenoid muscle with sympathetic preganglionic neurons." *Laryngoscope*. 112(8 Pt 1):1397-406. PMID: 11929937
Further Reading
- Can electrical stimulation help denervated muscles recover?(/articles/can-electrical-stimulation-help-denervated-muscles-recover)
- How does timing and intensity affect structural recovery?(/articles/how-does-the-timing-and-intensity-of-electrical-stimulation-in-home-based-fes-hbfes-affect-the-structural-recovery-of-lower-motor-neurons-and-muscle-fibres-following-a-complete-denervation-injury)
- Why your NMES product probably doesn't work with denervated muscle(/articles/why-your-nmes-product-probably-doesnt-work-with-denervated-muscle)
- Complete vs incomplete spinal cord injury: what you need to know(/articles/complete-vs-incomplete-spinal-cord-injury-what-you-need-to-know)